This content is sponsored by Passy-Muir
The number of adult and pediatric patients with tracheostomies are growing each year secondary to advancements in medical care and interventions to sustain life more so than seen historically. In a study conducted in 2008, it was estimated that by the year 2020, there would be over 600,000 adult patients requiring prolonged mechanical ventilation.[1] But little did that author know that, in 2020, a pandemic would change the face of medical care. It was estimated that in 2020, 965,000 people would require mechanical ventilation due to COVID-19, not including other disease and injury processes.[2] Just considering COVID-19 patients, the potential incidence of tracheostomies is thought to be much higher than the prediction given in 2008.
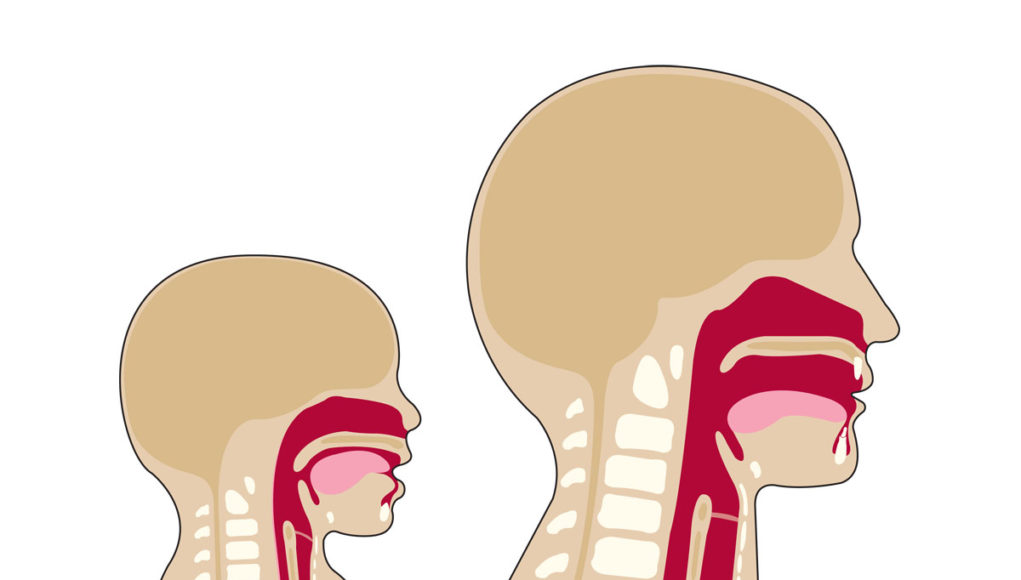
Aerodigestive changes following tracheostomy
With tracheostomies, changes in the aerodigestive system become evident through impacts on voice, swallowing, cough, and other functions. The prevalence for these aerodigestive challenges, which may lead to feeding and swallowing difficulties, is high.
The placement of a tracheostomy tube and prolonged mechanical ventilation with an inflated cuff causes a disconnect between the upper and lower airway. The lack of airflow through the upper airway can often lead to multiple negative changes affecting speech and swallowing: reduced subglottic pressure;[3] decreased sensation to the pharynx and glottis;3 reduced laryngopharyngeal reflex;[4] decreased ability to manage secretions, requiring more frequent suctioning;[5] decreased sense of taste and smell;[6] inability to vocalize; increased aspiration risk; and muscle disuse and atrophy.[7] A disconnect between respiration and swallowing also may negatively impact the ability to coordinate breathing and swallowing. For pediatrics, long term tracheostomy placement also has been associated with delayed acquisition of language, delayed social development, and risk of impaired parent-child bonding.[8],[9]

A primary means for closing the system to restore more normal physiology and pressures for patients with tracheostomies is the use of a bias-closed position, no-leak valve. When a patient has a tracheostomy, airflow is directed in and out through the tracheostomy tube and bypasses the upper airway. The Passy-Muir® Valve works by closing at the end of inspiration, which redirects 100% of airflow upwards through the vocal cords and upper airway. Research has shown that this redirection of airflow assists with improving secretion management, increasing sensory awareness, improving swallowing, and restoring natural physiologic PEEP (positive end- expiratory pressure), among other benefits.[10]
Normalizing function
Assessment and usage of a Valve also is important for the normalization of functions for all patients and for development in children. The primary consideration during assessment is that the patient has a patent airway, meaning the patient can exhale around the tracheostomy tube. Having a qualified team, familiar with airway management, is a key component of successful Valve use. The participation of infants, toddlers, and young children in the assessment process may be more difficult than with adults because of their limited ability to follow commands and volitionally vocalize; therefore, additional methods, such as transtracheal pressure (TTP) measurements, may be used to assess airway patency.[11] TTP is a method for measuring the pressure in the airway with the tracheostomy tube in place. It can be used with finger occlusion or a speaking valve to determine airway patency. TTP has been found to be a predictor associated with successful use of the Passy-Muir Valve (PMV). [12]
While speech and language development is an important consideration in pediatrics, research from the adult population suggests significant benefits for improved secretion management, cough function, and swallowing, all of which are influenced by pressure.1 Research has shown that subglottic pressure is reduced with a tracheostomy, impacting feeding and swallowing, cough, and secretion management.[13] Pullens and Streppel (2021) discussed the importance of restoring normal airway physiology to assist with feeding and swallowing, which would include restoring pressure, by using a speaking valve in the pediatric population. The adult population has several studies which indicate the need to restore subglottic pressure to assist with improved laryngeal function, swallowing, cough, and secretion management.1,[14]

The negative impact on pressures and the diminished stimulation of sensory receptors may affect feeding and swallowing in the pediatric population, to include oral-motor sensation. Henningfeld, Lang, and Goday (2019) reported that g-tube feeding and delayed feeding skills were associated with tracheostomy. They also hypothesized that children with tracheostomies would have more feeding issues than their age-matched peers without tracheostomies.[15] During review, they found that a history of ventilator-dependence, cuffed tracheostomy tube, and speaking valve use during inpatient care were inconsistently associated with later feeding and nutrition evaluations. However, the authors suggested that their findings also indicated that earlier speaking valve use has the potential to decrease later issues with feeding.
Early assessment for speaking valve use either in-line with mechanical ventilation or with a spontaneous breather leads to early intervention – in this case, establishing treatment plans, accommodations, and interventions earlier during their care . Early intervention and use of the PMV has been shown to have benefits with restoring the physiology of the upper airway to its more “normal” state by returning airflow through the upper airway during exhalation.7 This restoration of airflow to the upper airway allows evaluation of airway patency, vocal cord function, secretion management, swallowing, and communication skills.7 Research has shown that the use of a Passy Muir Valve can provide benefit during swallowing by increasing laryngeal excursion, returning cough and throat clear, and providing overall improved protection of the airway.7
Furthermore, patients on mechanical ventilation often experience psychosocial distress related to their inability to communicate with family and caregivers and to participate in their own care. Early implementation of the PMV increases the opportunity for patients to speak, swallow, and participate in direct therapy and to do so sooner. This early intervention has the potential to reduce anxiety, wean times, and lengths of stay. The restoration of communication and restoring oral nutrition have both been shown to have positive psychological benefits and to decrease anxiety, stress, fear, and other negative effects.[16]
Whitmore et al. (2020) also reported that the use of speaking valves for patients with and without mechanical ventilation was highly supported among the reviewed literature to promote speech and communication, which had an additional impact on patient satisfaction, and has been shown also to contribute to alveolar recruitment, weaning, and quality of life.[17] One barrier identified to using measurement tools with patients for assessing pain, cognitive status, and other areas while in the ICU is the ability of the patient to participate verbally.[18] Zaga et al. (2020) identified that the use of a one-way valve in-line with mechanical ventilation would assist with increasing the relevance of some measures.18
Intrathoracic and intra-abdominal pressures
Additional primary areas of pressure to consider when addressing the needs of patients with tracheostomies are the effects on the respiratory system and intrathoracic and intra-abdominal pressures, which also are diminished by having an open system.[19] With the redirection of airflow, the patient is no longer using the upper respiratory airway – airflow does not go through the upper airway and glottis (vocal cords). Use of the upper airway and glottis typically allows for control of exhalation and assists with controlling expiratory lung volumes.[20] This loss of pressure may impact gross motor function for mobility and postural stability.
Use of the Valve during physical therapy helps restore the pressure support in the trunk, allowing for natural increases in intrathoracic pressure (ITP) and intra-abdominal pressures (IAP) in response to increased postural demands. With an open tracheostomy tube and therefore, an open system, thoracic pressures cannot be increased or sustained as airflow passes through the tracheostomy tube and bypasses the upper airway. This difficulty would be observed when a patient needs to crawl, sit, push, or stand up. The typical means of gross motor movement for mobility is to engage the glottis (vocal cords) to restrict the expiratory lung volume to stabilize the chest and upper body.7,8 Placing a Passy Muir Valve on the tracheostomy tube closes the system and restores a patient’s ability to use the upper airway to control expiratory flow and improve ITP and IAP.
Consider that with infants and young children, a tracheostomy also could limit or diminish gross motor development. During infancy and early development, children are progressing through the stages of head control, trunk control, sitting, reaching, standing, and walking. Without good ITP and ITA, these functions could be significantly impacted and even delayed. A vicious cycle may begin as fine motor skills related to feeding, self-feeding, and other levels of function are directly linked to gross motor development. These delays and limitations can be mitigated by using a Passy Muir Valve to return the young child to a more normalized use of the upper airway with control of volumes and improved trunk control and postural stability. For adults, restoring pressure improves and restores functions that aid in recovery and quality of life.
Conclusion
The provision of multiple services for these patients with tracheostomies assists with overall care and recovery. Having these patients receive intervention to restore communication and swallowing, improves overall mental health which in turn impacts motivation and recovery.9 Taking into consideration the impacts disease processes and intubation or tracheostomies have on communication and swallowing, early assessments may be a key component to restoring patients’ abilities to communicate, eat, and return to more normal function, no matter the age.
*This is a sponsored post from Passy-Muir.
References
[1] Zilberberg, M. D., & Shorr, A. F. (2008). Prolonged acute mechanical ventilation and hospital bed utilization in 2020 in the United States: Implications for budgets, plant and personnel planning. BMC Health Services Research, 8(1), 242. https://doi.org/10.1186/1472-6963-8-242
[2] Halpern, N. A. and Tan, K. S. (2020). United States resource availability for COVID-19. Society for Critical Care Medicine, 3, 1 – 16. Retrieved from https://sccm.org/Blog/March-2020/United-States-Resource-Availability-for-COVID-19
[3] Eibling D & Gross R. (1996). Subglottic air pressure: a key component of swallowing efficiency. Annals of Otology, Rhinology & Laryngology, 105 (4), 253-258.
[4] Sasaki, C., Suzuki, M., Horiuchi, Masatoshi, H., & Kirchner, J.A. (1997). The effect of tracheostomy on the laryngeal closure reflex. The Laryngoscope, 87, 1428-1433.
[5] Siebens, A. A., Tippett, D. C., Kirby, N., & French, J. (1993). Dysphagia and expiratory air flow. Dysphagia, 8(3), 266-269. https://doi.org/10.1007/bf01354549
[6] O’Connor, L., Morris, N., & Paratz, J. (2019). Physiological and clinical outcomes associated with use of one-way speaking valves on tracheostomised patients: A systematic review. Heart & Lung, 8(4), 356-364. https://doi:10.1016/j.hrtlng.2018.11.006
[7] Griffiths R & Jones C. (1999). Recovery from intensive care. British Medical Journal, 319, 427.
[8] Cowell, J., Schlosser, D., & Joy, P. (2013). Language outcomes following infant tracheostomy. Asia Pacific Journal of Speech, Language, and Hearing, 5(3), 179 – 186. https://doi.org/10.1179/136132800805576942
[9] Lieu, J.E., Muntz, H.R., Prater, D., and Stahl, M.B. (1999). Passy-Muir valve in children with tracheotomy. International Journal of Pediatric Otorhinolaryngology, 50(3), 197-203. https://doi.org/10.1016/s0165-5876(99)00245-1
[10] O’Connor, L. R., Morris, N. R., & Paratz, J. (2019). Physiological and clinical outcomes associated with use of one-way speaking valves on tracheostomised patients: A systematic review. Heart Lung, 48(4), 356-364.https://doi:org/10.1016/j.hrtlng.2018.11.006
[11] Abraham, S. (2018). Candidacy for Passy Muir® Valve placement in infants and young children: The airway assessment. Aerodigestive Health, 1(1), 12-13.
[12] Brooks, L., Figueroa, J., Edwards, T., Reeder, W., McBrayer, S., & Landry, A. (2020). Passy muir valve tolerance in medically complex infants and children: Are there predictors for success? The Laryngoscope, 130(11), E632 – E939. https://doi.org/10.1002/lary.28440
[13] Pullens, B. & Streppel, M. (2021). Swallowing problems in children with tracheostomies. Seminars in Pediatric Surgery, 30, 151053. https://doi.org/10.1016/j.sempedsurg.2021.151053
[14] Alhashemi, H., Algarni, M., Al-Hakami, H., Seebran, N., Hussain, T., Bhutto, T., Tashkandi, A., Alayed, M., Bukhari, E., & Alzahari, A. (2022). An interdisciplinary approach to the management of individuals with tracheostomy. Respiratory Care, 67(1), 34 – 39. https://doi.org/10.4187/respcare.08869
[15] Henningfeld, J., Lang, C., and Goday, P. (2019). Feeding disorders in pediatric tracheostomy. American Journal of Respiratory and Critical Care Medicine, 199: A3583. https://doi.org/10.1164/ajrccm-conference.2019.199.1_MeetingAbstracts.A3583
[16] Freeman-Sanderson, A. L., Togher, L., Elkins, M .R., & Phipps, P. R. (2016). Return of voice for ventilated tracheostomy patients in ICU: A randomized, controlled trial of early-targeted intervention. Critical Care Medicine, 44(6), 1075-1081. https://doi.org/10.1097/ccm.0000000000001610
[17] Whitmore, K.A., Townsend, S.C., & Laupland, K.B. (2020). Management of tracheostomies in the intensive care unit: a scoping review. BMJ Open Respiratory Research, 7(1).
[18] Zaga, C.J., Cigognini, B., Vogel, A.P., & Berney, S. (2020). Outcome measurement tools for communication, voice and speech intelligibility in the ICU and their clinimetric properties: A systematic review. Journal of Intensive Care Society, 0(0), 1 – 14. https://doi.org/10.1177/1751143720963757
[19] Massery, M. (2014). Expert interview: The role of the Passy-Muir valve in physical therapy. Talk Muir (3 Feb. 2014), 2-4.
[20] Massery, M., Hagins, M., Stafford, R., Moerchen, V., & Hodges, V. W. (2013). Effects of airway control by glottal structures on postural stability. Journal of Applied Physiology, 115(4), 483-490.